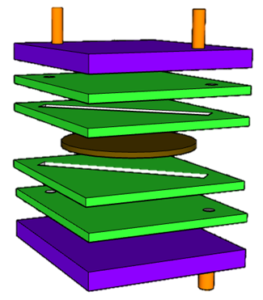
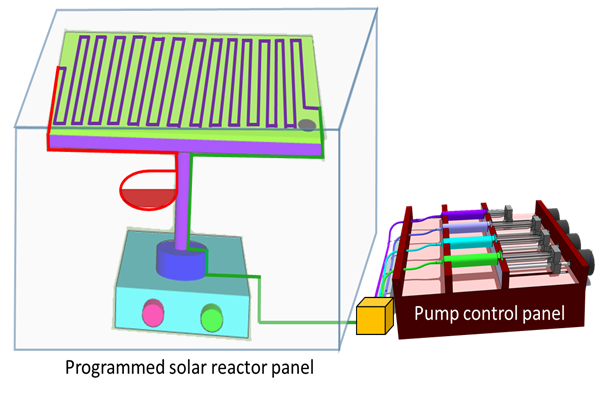
Our SCS-L Flow/Batch Reactor is an exemplary multi-purpose reactor and specially designed for learning and education purpose. Allows user to cover a wide range of chemical applications. This reactor is user friendly, well designed for the beginners, who are entering into the flow/batch chemistry field. The SCS-L reactor enable its user to perform homogeneous, heterogeneous, high temperature reactions, electrochemical and photochemical reactions as well.
Specifications of Learning Multipurpose Batch/Flow Reactor :
- Reactor volume: 5 ml (rectangular) for continuous flow
- Inert wetted materials: PTFE sheet
- Reactor body- Bakelite sheet
- pressure: 2-3 bar
- Optional (batch reactor with 20 ml volume)
- Syringe pump with variable flow rate:
- Nano liter to several milliliter/min.
- Control unit for backward, forward, slow, fast etc.
- Plated MS heater with temperature range (240W): RT – 150 o
- Control panel- Nylon
- Power supply for electrochemistry with fine tuning:
- Variable current (0-2A), variable voltage (0-29 V).
- Fix high current and low volt (5A and 12V)
- Fix low current and high voltage (2A and 110V)
- Electrodes: Graphite
- optional: Zinc, Carbon, Copper, Aluminium, Platinum, Stainless Steel mess, Iron, Nickle, Titanium etc)
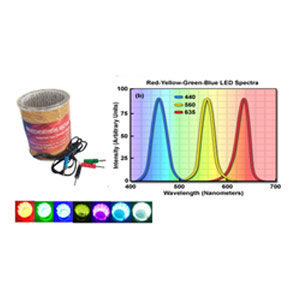
- Photochemical reactor (30W LED): Red, Blue, Green, Yellow, Cyan, White light etc.
Some more details of the Smartchemsynth Learning Flow Reactor (SCS-L)
Photochemistry
The use of this reactor for photochemical applications can overcome most of the issues associated with batch photochemistry. Provides opportunities to ensure a uniform irradiation of the entire reaction mixture. Photochemical reactions are driven by the number of photons that are able to activate molecules causing the desired reaction. Photochemical multiphasic reactions completely exploit the photon- and mass-transfer enhancement properties offered by flow chemistry. Advantages of photochemistry enhanced energy efficiencies and reductions of solvent volumes and consequently waste. The typical setup for photochemical processes comprises transparent body made of acrylic, efficiently coiled with a light source compromising of different colours (wavelength) of light.
Electrochemistry
In continuous flow process, the transfer of electrons drives reactions and transformations, meaning that electrochemistry is a surface phenomenon whereby reactions are optimised when there is a high surface area to volume ratio. The reduced distance between the electrodes allows better control of the number of electrons transferred to the reaction media, enabling better control and selectivity. It is possible to generate reactive species without the need for hazardous oxidizing and reducing agents.
It is well known that the nature of the electrode material and the over-potential accessible with certain electrodes influence the electrochemical processes. Fine-tuning of electrodes can be very important in order to optimize reactions. Therefore, manipulating electrochemistry in flow gives possible degree of flexibility since the application has control on the flow parameters, the nature of the electrochemical measurement, on the dimensions and nature of the electrode. Benefits of electrochemistry are rapid oxidations and reductions-oxidative synthesis of drug metabolites and oxidative synthesis of drug metabolites.
High Temperature Reactions
There are many chemical reactions in chemistry which need high temperature to reach the desired conversions or yield, till now with traditional batch equipment it is tough, whereas with flow chemistry we can overcome this problem in a very significant way. On increasing the temperature, the rate of reaction increases for many reactions, taking place at around room temperature, and the rate of reaction doubles for every 10 0C rise in temperature.
Homogeneous and Heterogeneous Reactions
The homogeneous & heterogeneous in continuous process offers preferable process control, higher efficiency, and comparable or superior reaction profiles and yields compared to batch conditions.
Applications of Learning Multipurpose Batch/Flow Reactor
- Metal free oxidation
- Electricity as green oxidant/reductant
- Ozonolysis
- Gas-liquid reaction,
- Electrolysis at high temperature
- Halogenation
- Nitration
- Grignard reaction mechanisms
- Lithiation reaction
- Reactions that use toxic gases
- Diazomethane